 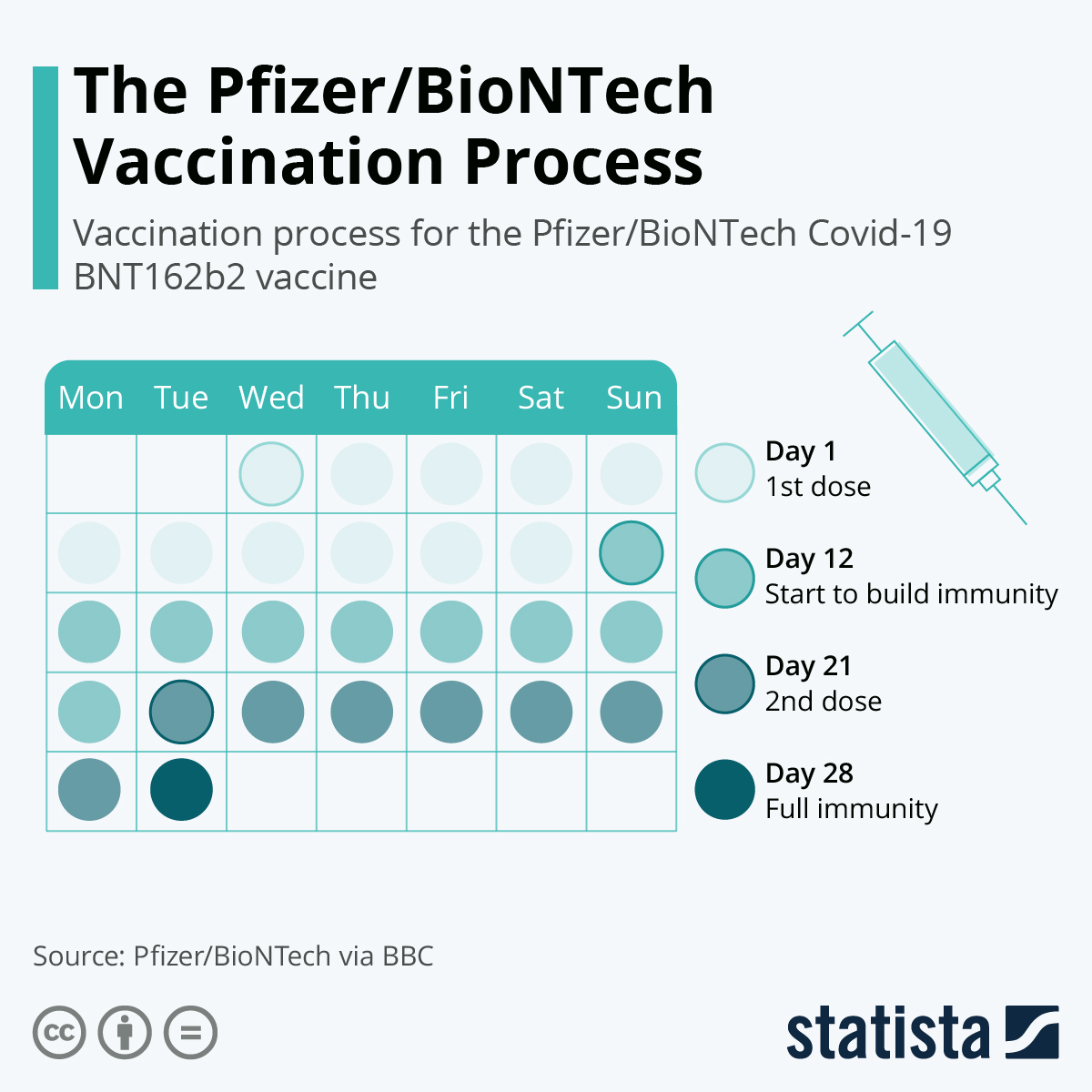
Previously received an incomplete series of Original monovalent or bivalent mRNA vaccine doses: Complete the vaccination series with 1 or 2 homologous updated (2023–2024 Formula) mRNA vaccine doses, depending on vaccine manufacturer and the number of previous vaccine doses.Unvaccinated: 2 or 3 homologous (i.e., from the same manufacturer) updated (2023–2024 Formula) mRNA vaccine doses, depending on vaccine manufacturer (i.e., Moderna, Pfizer-BioNTech).The recommended vaccine type and number of updated (2023–2024 Formula) COVID-19 vaccine doses are based on age and vaccination history. The COVID-19 vaccination schedule for people who are not moderately or severely immunocompromised is detailed in Table 1. Vaccine doses should be administered by the intramuscular route. However, for children who transition from age 4 years to age 5 years and children who are moderately or severely immunocompromised and transition from age 11 years to age 12 years, FDA authorization allows for an alternative dosage (see Table 1, Table 2, and Transitioning from a younger to older age group). Vaccine dosage and administrationĬDC recommends that people receive the age-appropriate vaccine product and dosage based on their age on the day of vaccination and in accordance with the recommended intervals for that age group ( 1 ). See Appendix A for recommendations for people who received COVID-19 vaccine outside the United States. Vaccination schedules can be found in Table 1 for people who are not moderately or severely immunocompromised and in Table 2 for people who are moderately or severely immunocompromised. There is no preferential recommendation for the use of any one COVID-19 vaccine over another when more than one recommended and age-appropriate vaccine is available. CDC recommends that people stay up to date with COVID-19 vaccination. 
There is currently no FDA-approved or FDA-authorized COVID-19 vaccine for children younger than age 6 months. COVID-19 Vaccine Product Information can be consulted for a full list of ingredients and information on the conditions of use, storage and handling, preparation, and administration procedures.ĬOVID-19 vaccination is recommended for everyone ages 6 months and older in the United States for the prevention of COVID-19. The Original monovalent and bivalent (Original and Omicron BA.4/BA.5) formulations should no longer be used.ĬOVID-19 vaccine-specific package inserts and FDA fact sheets and U.S. 
The 2023–2024 formulation for all COVID-19 vaccines licensed or authorized in the United States (Moderna, Novavax, and Pfizer-BioNTech) has been updated to a monovalent vaccine based on the Omicron XBB.1.5 sublineage of SARS-CoV-2.Novavax COVID-19 Vaccine, Adjuvanted (hereafter referred to as Novavax COVID-19 Vaccine) is authorized for people ages 12 years and older.These vaccines are hereafter referred to as updated (2023–2024 Formula) Pfizer-BioNTech COVID-19 Vaccine. Pfizer-BioNTech COVID-19 Vaccine (2023–2024 Formula) is authorized for children ages 6 months–11 years COMIRNATY is the licensed Pfizer-BioNTech product for people ages 12 years and older.These vaccines are hereafter referred to as updated (2023–2024 Formula) Moderna COVID-19 Vaccine. Moderna COVID-19 Vaccine (2023–2024 Formula) is authorized for children ages 6 months–11 years SPIKEVAX is the licensed Moderna product for people ages 12 years and older.Two types of COVID-19 vaccines are available for use in the United States: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
